Cigar Science is an occasional feature of Cigarfan.net, an occasion which is usually prompted by the common cold or flu. In lieu of a cigar review, this is my humble effort to identify the organoleptic properties of cigar tobacco and to determine if the outlandish olfactory findings of certain cigar reviewers are justified… or if we should just keep our hallucinations to ourselves.
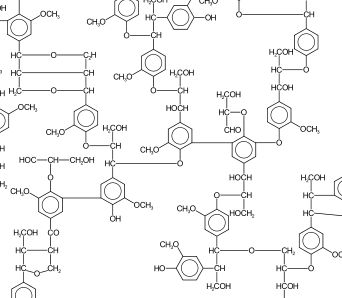
Lignin is an essential element of wood, and of most vascular plants, including tobacco. (The word itself is derived from the Latin lignum, meaning wood.) Lignin fills the spaces between the cell walls of many plants, provides strength, and is thought to be responsible for moving water through cell structures.
This would be of no interest to cigar smokers except for one fact: lignin is “the most abundant natural organic aromatic polymer found in the vascular plant kingdom.”¹ Lignin accounts for 4 to 5 percent of tobacco weight; far less than hard wood, but significant nevertheless.
When burned, lignin produces phenols with specific aromatic properties, the most evident of which is the smell of wood burning. Two of these phenols, guaiacol and syringol, are responsible for the flavors of smoked foods. That is one of the reasons why more highly lignified woods (hardwoods) are preferred for barbeque.
Two of these phenols, guaiacol and syringol, are responsible for the flavors of smoked foods. That is one of the reasons why more highly lignified woods (hardwoods) are preferred for barbeque.
 Guaiacol is associated with smoky flavor, and is present in roasted coffee compounds, while syringol is associated with the aroma of wood smoke. Guaiacol is also a precursor to other phenolic compounds that contribute to flavors commonly found in cigar smoke: vanillin and eugenol. We all know what vanillin is, but the term eugenol is derived from the scientific name for clove, and its aroma is exactly that: clove-like. It has also been extracted as an essential oil from nutmeg, cinnamon, and basil.
Guaiacol is associated with smoky flavor, and is present in roasted coffee compounds, while syringol is associated with the aroma of wood smoke. Guaiacol is also a precursor to other phenolic compounds that contribute to flavors commonly found in cigar smoke: vanillin and eugenol. We all know what vanillin is, but the term eugenol is derived from the scientific name for clove, and its aroma is exactly that: clove-like. It has also been extracted as an essential oil from nutmeg, cinnamon, and basil.
Other phenols produced include cresols, which have a petroleum-like scent. Cresols are also found in coal tar, crude oil, and creosote. Cresols are also, apparently, a component of Sharpie markers. I’m already on the lookout for a cigar review that includes “a whiff of Sharpie marker.”
These phenolic constituents are also produced as pheromones by certain insects. The desert locust is ardently attracted to guaiacol, and the orchid bee is erotically aroused by both eugenol and p-cresol. So don’t be surprised if these guys crash your next herf. They only want to make friends.
It’s difficult to say what chemical compounds are specifically responsible for the flavors and aromas of cigar tobacco –there are thousands of them — but it seems reasonable to me to say that we aren’t hallucinating when we find vanilla, or clove, or related aromas in cigar smoke. Some cigar smokers are more sensitive to subtle aromas than others, and some people are just better at putting names to sensations; but I don’t think that it’s fair to say that an unexpected flavor, like honey or aniseed, is the product of an overactive imagination. Different people won’t always agree on the flavors they find in a particular blend, but to deny that those flavors are there seems unwarranted when there is a basis for it in chemical analysis.
Other flavor and aroma compounds have been discussed in previous articles, available here.
Notes and Selected References
1. Leffingwell, J. (2001) Chemical constituents of tobacco leaf and differences among tobacco types. Leffingwell Reports, 1(2). Retrieved from: http://www.leffingwell.com/leffrept.htm
2. McGee, H. (2004) On food and cooking: the science and lore of the kitchen. New York: Scribner.
3. Hardwood Smoke Science. Steel Town BBQ. retrieved from: http://steeltownbbq.com/7.html